Research
The cell shape and the rigidity of the extra-cellular environment have been shown to play an important role in the regulation of cellular processes such as cell division and differentiation; they have also been shown to play an important role in the development of tissues and in the onset of cancer and other pathological states and diseases. We study the physical principles that govern the response of cells to mechanical cues in their surroundings using elasticity theory and condensed matter physics. This includes phenomena such as cell adhesion, establishment of cell shape and cytoskeleton structure, elastic interactions of cells, patterning, aggregation and alignment of cells. Using computer simulations we also investigate the interaction of motor proteins and cytoskeleton filaments that produce forces and movements of cells including, stress-fiber assembly, axonal growth and blood platelet formation. For more information, see Table of Contents on the Left.
For news coverage of our work on the mechanics of stem cells go here: Hebrew University, Ynet
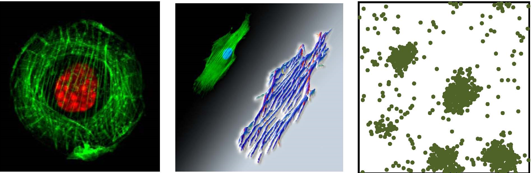
Cell Matrix Interactions
How do the mechanical properties of the environment influence cell morphology and fate?
Cell adhesion is a critical stage in the life of adherent cells such as muscle cells, epithelial cells and stem cells. Upon binding to the extra-cellular matrix, elastic stresses are actively generated in the cell by numerous molecular motors (mainly myosin) that locally compress (and globally stretch) the cytoskeleton. By pulling on the environment cells can 'sense' and respond to mechanical properties of their surroundings (for instance the rigidity and topography of the extracellular matrix or the presence of other contractile cells in their vicinity). Indeed, experiments have shown that elastic stresses that develop in the cell cytoskeleton upon cell adhesion feedback on myosin motor activity governing their assembly with actin filaments to form organized muscle-like fibers, called stress-fibers. This creates a mechanical feedback loop between the cell and the environment that affects the morphology, internal structure and eventually the fate of the cell. Using elasticity theory and principles of condensed matter physics we study the dynamics and energetics of cell-matrix interactions, including the reorganization and formation of stress-fibers in the cytoskeleton, anchorage to the extracellular matrix and the accompanying development of the cell shape, both in stationary and migrating cells. To read more about this see the references below. Ref. [1-3] discuss the effects of substrate rigidity and cell shape on the development of acto-myosin stress fibers in cells. Ref [4] analyses the effects of the cell nucleus on the orientation of actomyosin stress fibers around it. Ref. [5-6] present a simple mechanical model of cell spreading dynamics.

Also see news coverage of our work on the mechanics of stem cells here: Hebrew University, Ynet
- Zemel, A, F Rehfeldt, AEX Brown, DE Discher, and SA SAFRAN. 2010. “Optimal Matrix Rigidity For Stress-Fibre Polarization In Stem Cells.” Nature Physics 6. 468–473.
- Zemel, A, F Rehfeldt, AEX Brown, DE Discher, and SA SAFRAN. 2010. “Cell Shape, Spreading Symmetry, And The Polarization Of Stress-Fibers In Cells.” Journal Of Physics: Condensed Matter 22. IOP Publishing: 194110.
- Nisenholz, Noam, Mordechai Botton, and Assaf Zemel. 2014. “Early-Time Dynamics Of Actomyosin Polarization In Cells Of Confined Shape In Elastic Matrices.” Soft Matter 10. Royal Society of Chemistry: 2453–2462.
- Zemel, Assaf. 2015. “Active Mechanical Coupling Between The Nucleus, Cytoskeleton And The Extracellular Matrix, And The Implications For Perinuclear Actomyosin Organization.” Soft Matter 11. Royal Society of Chemistry: 2353–2363.
- Nisenholz, Noam, Kavitha Rajendran, Quynh Dang, Hao Chen, Ralf Kemkemer, Ramaswamy Krishnan, and Assaf Zemel. 2014. “Active Mechanics And Dynamics Of Cell Spreading On Elastic Substrates.” Soft Matter 10. Royal Society of Chemistry: 7234–7246.
- Nisenholz, Noam, Aishwarya Paknikar, Sarah Köster, and Assaf Zemel. 2016. “Contribution Of Myosin Ii Activity To Cell Spreading Dynamics.” Soft Matter 12. Royal Society of Chemistry: 500–507.
Elastic Interactions of Cells
In what ways do the forces generated locally by cells affect other cells in their environment?
Mechanical forces exerted by cells not only feedback on the morphology and internal structure of each cell individually, but may also impact other cells in the surroundings. These, generally long-ranged elastic interactions are thought to play an important role in the orientation and self-assembly of cells to form organized tissue structures. We develop tools based on generic considerations of force balance to predict how cells actively self-assemble and orient to form organized tissue structures by virtue of the forces they exert on the matrix and their response to these forces. Our models take into account the active forces exerted by the cells, the elastic resisting forces of the environment, as well as the feedback mechanisms that modulate cell activity. This results in a complicated self-consistency problem to solve for the force generation and self-assembly of cells in the medium. Interestingly, because elastic interactions are long-ranged, some aspects of cell-cell elastic interactions may be understood by analogy to other systems with long-ranged interactions such as electrical dipoles in a dielectric. The slide below shows how a theory for the active susceptibility of cells to externally applied forces was developed by analogy to the theory of polar dielectrics developed by Lars Onsager in the 30's. To read more on this interesting analogy see the references below.
In addition, we develop computer simulations and approximate analytical theory to study the self-assembly properties of ensembles of elastically interacting cells. One interesting prediction of our analysis is the regulation of cellular self-assembly behavior by the rigidity of the surrounding extracellular matrix. We predict how the surroundings' rigidity may alter the propensity of cells to aggregate, disperse or form interesting phases depending on their density and inherent stochastic nature.

- . 2006. “Active Elasticity Of Gels With Contractile Cells.” Physical Review Letters 97. APS: 128103.
- . 2007. “Active Self-Polarization Of Contractile Cells In Asymmetrically Shaped Domains.” Physical Review E 76. APS: 021905.
Collective Interactions of Motor Proteins and Filaments in the Cytoskeleton
How do motor proteins produce order in the cytoskeleton of cells?
Interactions of multiple molecular motors with actin and microtubule filaments form the basis of many cytoskeletal processes including axonal growth, muscle contraction, cell division and platelet formation. We develop computer simulations to study, on the molecular level, how motor proteins and cytoskeletal filaments generate forces and interact spontaneously to form organized structures in the cytoskeleton. Our simulations allow us to study the dynamics of self-organization in large assemblies of filaments, comprising different types of motors at different densities and subject to different boundary conditions.

To read more on this research see:
- . 2008. “Expansion And Polarity Sorting In Microtubule-Dynein Bundles.” Progress Of Theoretical Physics Supplement 173. Oxford University Press: 17–25.
- . 2015. “Force Generation By Molecular-Motor-Powered Microtubule Bundles; Implications For Neuronal Polarization And Growth.” Frontiers In Cellular Neuroscience 9: 441.
Mechanics of Neuron Growth and Differentiation
What are the forces driving the growth and maturation of axons and dendrites in neurons?
Axons and dendrites, the prominent structures emanating from the cell body of neurons, are the processes responsible for the transmission and reception of electrical signals through the nervous system. While the structure of individual neurons is a key to the function of a neuronal network, still very little is known on how it develops on the mechanical and molecular levels. A major factor in the cytoskeleton of axons and dendrites are dense bundles of microtubules that run across their length, providing tracks for molecular motors to transmit vesicles filled by nutrients and other factors from the cell body outwards and vice versa. In addition, those heavily dense microtubule bundles and the molecular motors that bind them, are believed to play a key role in driving the growth and maturation of neurons. The sliding motion of molecular motors along microtubules not only drives the filaments' motion but also contributes to their sorting along the process. However, a quantitative understanding of how the ensembles of molecular motors and cytoskeleton filaments cooperate to produce forces and structure in neuronal processes is still lacking. To investigate these questions we develop computer simulations that allow us to calculate the dynamics of filaments in bundles of microtubules that are cross-linked and powered by molecular motors. The motion of filaments and the forces they exert are investigated as a function of the motor type, the microtubule density and length, applied load and motor connectivity. The work is carried out in close collaboration with the experimental group of Kristian Franze at Cambridge University.
To read more about this project see:
Jakobs, Maximilian, Kristian Franze, and Assaf Zemel. 2015. “Force Generation By Molecular-Motor-Powered Microtubule Bundles; Implications For Neuronal Polarization And Growth.” Frontiers In Cellular Neuroscience 9: 441
Jakobs, Maximilian, Kristian Franze, and Assaf Zemel. 2020. “Mechanical Regulation of Neurite Polarization and Growth: A Computational Study.” Biophysical Journal, in press
