Citation:
Masarwa, A. ; Fürstnerb, A. ; Marek*, I. Metal-Catalyzed Rearrangement of enantiomerically Pure Alkylidenecyclopropane Derivatives as a New access to Cyclobutenes Possessing Quaternary stereocenters. Chem. Commun. 2009, 5760-5762.
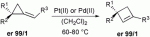
Abstract:
Pd(II)- and Pt(II)-catalyzed ring-expansion of enantiomerically pure alkylidenecyclopropane derivatives leads to the formation of cyclobutene species with a complete preservation of the stereogenic center.

