Citation:
I. Alshanski, D. E. Shalev, S. Yitzchaik, and M. Hurevich. 2021. “Determining the structure and binding mechanism of oxytocin-Cu(2+) complex using paramagnetic relaxation enhancement NMR analysis.” J Biol Inorg Chem.
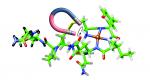
Abstract:
Oxytocin is a neuropeptide that binds copper ions in nature. The structure of oxytocin in interaction with Cu2+ was determined here by NMR, showing which atoms of the peptide are involved in binding. Paramagnetic relaxation enhancement NMR analyses indicated a binding mechanism where the amino terminus was required for binding and subsequently Tyr2, Ile3 and Gln4 bound in that order. The aromatic ring of Tyr2 formed a π-cation interaction with Cu2+. Oxytocin copper complex structure revealed by paramagnetic relaxation enhancement NMR analyses.


