Sharir A, Stern T, Rot C, Shahar R, Zelzer E.
Muscle force regulates bone shaping for optimal load-bearing capacity during embryogenesis. DEVELOPMENT [Internet]. 2011;138 (15) :3247-3259.
Publisher's VersionAbstractThe vertebrate skeleton consists of over 200 individual bones, each with its own unique shape, size and function. We study the role of intrauterine muscle-induced mechanical loads in determining the three-dimensional morphology of developing bones. Analysis of the force-generating capacity of intrauterine muscles in mice revealed that developing bones are subjected to significant and progressively increasing mechanical challenges. To evaluate the effect of intrauterine loads on bone morphogenesis and the contribution of the emerging shape to the ability of bones to withstand these loads, we monitored structural and mineral changes during development. Using daily micro-CT scans of appendicular long bones we identify a developmental program, which we term preferential bone growth, that determines the specific circumferential shape of each bone by employing asymmetric mineral deposition and transient cortical thickening. Finite element analysis demonstrates that the resulting bone structure has optimal load-bearing capacity. To test the hypothesis that muscle forces regulate preferential bone growth in utero, we examine this process in a mouse strain (mdg) that lacks muscle contractions. In the absence of mechanical loads, the stereotypical circumferential outline of each bone is lost, leading to the development of mechanically inferior bones. This study identifies muscle force regulation of preferential bone growth as the module that shapes the circumferential outline of bones and, consequently, optimizes their load-bearing capacity during development. Our findings invoke a common mechanism that permits the formation of different circumferential outlines in different bones.
 muscle_force_regulates_bone_shaping_for_optimal_load-bearing_capacity_during_embryogenesis.pdf
muscle_force_regulates_bone_shaping_for_optimal_load-bearing_capacity_during_embryogenesis.pdf Sharir A, Israeli D, Milgram J, Currey JD, Monsonego-Ornan E, Shahar R.
The canine baculum- The structure and mechanical properties of an unusual bone. JOURNAL OF STRUCTURAL BIOLOGY [Internet]. 2011;175 (3) :451-456.
Publisher's VersionAbstractThe baculum is an extraskeletal bone located in the penis of a few species in several orders of mammals such as carnivores, insectivores, rodents, bats and primates.
This study aims to describe the structure, architecture and mechanical properties of the canine baculum. To this end canine bacula from castrated and uncastrated dogs were collected and examined by light microscopy, micro-computed tomography (microCT) scanning, histological staining, and mechanical testing. Their mineral density and mechanical properties were compared with those of a typical skeletal bone (the radius) in the same dog. Furthermore, a numerical model of a representative baculum was created and its mechanical performance analyzed using the finite element method, in order to try to elucidate its function.
Examination of light microscopy images of transverse sections shows that the baculum consists of a typical sandwich structure, with two cortical plates separated, and joined, by loose cancellous bone. MicroCT scans reveal that the mineral density is lower in the baculum than in the radius, both in castrated as well as in uncastrated dogs, resulting in much lower stiffness. Castration was found to decrease the mineral density in both the baculum and the radius.
The most likely function of the baculum of the dog is to stiffen the penis to assist intromission, and its much lower mineral density compared to that of the radius may be a mechanism designed to decrease the stiffness somewhat, and thus reduce the risk of fracture during copulation. (C) 2011 Elsevier Inc. All rights reserved.
 the_canine_baculum-_the_structure_and_mechanical_properties_of_an_unusual_bone.pdf
the_canine_baculum-_the_structure_and_mechanical_properties_of_an_unusual_bone.pdf Idelevich A, Kerschnitzki M, Shahar R, Monsonego-Ornan E.
1,25(OH)2D3 Alters Growth Plate Maturation and Bone Architecture in Young Rats with Normal Renal Function. PLOS ONE [Internet]. 2011;6 (6).
Publisher's VersionAbstractWhereas detrimental effects of vitamin D deficiency are known over century, the effects of vitamin D receptor activation by 1,25(OH)(2)D-3, the principal hormonal form of vitamin D, on the growing bone and its growth plate are less clear. Currently, 1,25(OH)(2)D-3 is used in pediatric patients with chronic kidney disease and mineral and bone disorder (CKD-MBD) and is strongly associated with growth retardation. Here, we investigate the effect of 1,25(OH)(2)D-3 treatment on bone development in normal young rats, unrelated to renal insufficiency. Young rats received daily i.p. injections of 1 mu g/kg 1,25(OH)(2)D-3 for one week, or intermittent 3 mu g/kg 1,25(OH)(2)D-3 for one month. Histological analysis revealed narrower tibial growth plates, predominantly in the hypertrophic zone of 1,25(OH)(2)D-3-treated animals in both experimental protocols. This phenotype was supported by narrower distribution of aggrecan, collagens II and X mRNA, shown by in situ hybridization. Concomitant with altered chondrocyte maturation, 1,25(OH)(2)D-3 increased chondrocyte proliferation and apoptosis in terminal hypertrophic cells. In vitro treatment of the chondrocytic cell line ATDC5 with 1,25(OH)(2)D-3 lowered differentiation and increased proliferation dose and time-dependently. Micro-CT analysis of femurs from 1-week 1,25(OH)(2)D-3-treated group revealed reduced cortical thickness, elevated cortical porosity, and higher trabecular number and thickness. 1-month administration resulted in a similar cortical phenotype but without effect on trabecular bone. Evaluation of fluorochrome binding with confocal microscopy revealed inhibiting effects of 1,25(OH)(2)D-3 on intracortical bone formation. This study shows negative effects of 1,25(OH)(2)D-3 on growth plate and bone which may contribute to the exacerbation of MBD in the CKD pediatric patients.
 125oh2d3_alters_growth_plate_maturation_and_bone_architecture_in_young_rats_with_normal_renal_function.pdf
125oh2d3_alters_growth_plate_maturation_and_bone_architecture_in_young_rats_with_normal_renal_function.pdf Chattah NL-T, Kupczik K, Shahar R, Hublin J-J, Weiner S.
Structure-function relations of primate lower incisors- a study of the deformation of Macaca mulatta dentition using electronic speckle pattern interferometry (ESPI). JOURNAL OF ANATOMY [Internet]. 2011;218 (1) :87-95.
Publisher's VersionAbstractTeeth adopt a variety of different morphologies, each of which is presumably optimized for performing specific functions during feeding. It is generally agreed that the enamel cap is a crucial element in controlling the mechanical behavior of mammalian teeth under load. Incisors are particularly interesting in terms of structure-function relations, as their role in feeding is that of the 'first bite'. However, little is known how incisor cap morphology is related to tooth deformation. In the present paper we examine the mechanical behavior of mandibular central incisors in the cercopithecine primate Macaca mulatta under loads similar to those encountered during ingestion. We map three-dimensional displacements on the labial surface of the crown as it is compressed, using electronic speckle pattern interferometry (ESPI), an optical metrology method. In addition, micro-computed tomography is used to obtain data regarding the morphology of the enamel cap, which in the M. mulatta lower incisors exhibits missing or very little enamel on the lingual face. The results showed that although compressed along a longitudinal axis, deformation in the incisors mostly occurred in the lingual direction and orthogonal to the direction of the applied load. Both isolated, embedded teeth and teeth in the mandible showed considerable lingual deformation. Incisor deformation in the mandible was generally greater, reflecting the additional freedom of movement enabled by the supporting structures. We show that the association with adjacent teeth in the arch is significant for the behavior of the tooth under load. Finally, loading two teeth simultaneously in the mandible showed that they work as one functional unit. We suggest that these results demonstrate the importance of enamel cap morphology in directing deformation behavior; an ability stemming from the stiffness of the enamel cap overlying the more pliable dentin.
 structure-function_relations_of_primate_lower_incisors-_a_study_of_the_deformation_of_macaca_mulatta_dentition_using_electronic_speckle_pattern_interferometry_espi.pdf
structure-function_relations_of_primate_lower_incisors-_a_study_of_the_deformation_of_macaca_mulatta_dentition_using_electronic_speckle_pattern_interferometry_espi.pdf Kerschnitzki M, Wagermaier W, Roschger P, Seto J, Shahar R, Duda GN, Mundlos S, Fratzl P.
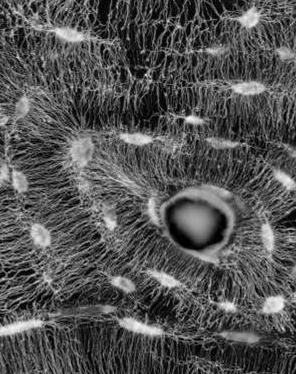
The organization of the osteocyte network mirrors the extracellular matrix orientation in bone. JOURNAL OF STRUCTURAL BIOLOGY [Internet]. 2011;173 (2) :303-311.
Publisher's VersionAbstractBone is a dynamic tissue that is continually undergoing a process of remodeling - an effect due to the interplay between bone resorption by osteoclasts and bone formation by osteoblasts. When new bone is deposited, some of the osteoblasts are embedded in the mineralizing collagen matrix and differentiate to osteocytes, forming a dense network throughout the whole bone tissue. Here, we investigate the extent to which the organization of the osteocyte network controls the collagen matrix arrangement found in various bone tissues. Several tissue types from equine, ovine and murine bone have been examined using confocal laser scanning microscopy as well as polarized light microscopy and back-scattered electron imaging. From comparing the spatial arrangements of unorganized and organized bone, we propose that the formation of a highly oriented collagen matrix requires an alignment of osteoblasts whereby a substrate layer provides a surface such that osteoblasts can align and, collectively, build new matrix. Without such a substrate, osteoblasts act isolated and only form matrices without long range order. Hence, we conclude that osteoblasts synthesize and utilize scaffold-like primary tissue as a guide for the deposition of highly ordered and mechanically competent bone tissue by a collective action of many cells. (C) 2010 Elsevier Inc. All rights reserved.
 the_organization_of_the_osteocyte_network_mirrors_the_extracellular_matrix_orientation_in_bone.pdf
the_organization_of_the_osteocyte_network_mirrors_the_extracellular_matrix_orientation_in_bone.pdf