Citation:
Abstract:
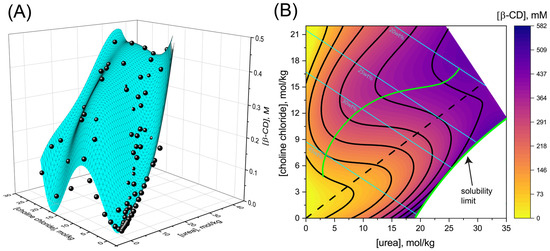
Deep eutectic solvents (DESs) show promise in pharmaceutical applications, most prominently as excellent solubilizers. Yet, because DES are complex multi-component mixtures, it is challenging to dissect the contribution of each component to solvation. Moreover, deviations from the eutectic concentration lead to phase separation of the DES, making it impractical to vary the ratios of components to potentially improve solvation. Water addition alleviates this limitation as it significantly decreases the melting temperature and stabilizes the DES single-phase region. Here, we follow the solubility of β-cyclodextrin (β-CD) in DES formed by the eutectic 2:1 mole ratio of urea and choline chloride (CC). Upon water addition to DES, we find that at almost all hydration levels, the highest β-CD solubility is achieved at DES compositions that are shifted from the 2:1 ratio. At higher urea to CC ratios, due to the limited solubility of urea, the optimum composition allowing the highest β-CD solubility is reached at the DES solubility limit. For mixtures with higher CC concentration, the composition allowing optimal solvation varies with hydration. For example, β-CD solubility at 40 wt% water is enhanced by a factor of 1.5 for a 1:2 urea to CC mole ratio compared with the 2:1 eutectic ratio. We further develop a methodology allowing us to link the preferential accumulation of urea and CC in the vicinity of β-CD to its increased solubility. The methodology we present here allows a dissection of solute interactions with DES components that is crucial for rationally developing improved drug and excipient formulations.
Notes: